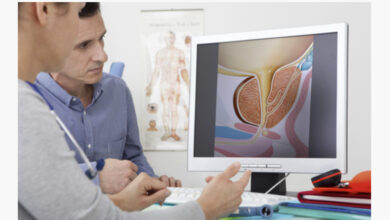
A REPLACEMENT FOR PSA: HAVE A PCA3 BEFORE PROSTATE BIOPSY

No blood test has been under more intense scrutiny than the beleaguered Prostate Specific Antigen (PSA). Approved by the FDA in 1986, PSA has been used by physicians everywhere to diagnose and monitor men with prostate cancer (CaP). In the U.S., CaP is the second most common non-skin malignancy among men and is the second leading cause of cancer death in men. African American men have a 1.7 times higher incidence of CaP and a 2.1 times higher rate of mortality from CaP than Caucasian men. American men have come to rely on a PSA test as an annual screening procedure for detection of CaP; despite several preventive services organizations recommending against its use, it is still heavily relied upon by American men.
The United States Preventative Services Task Force (USPSTF), the quasi-governmental recommendations committee, thinks PSA testing leads to unnecessary prostate biopsies so for men ages 55-69 they recommend consulting with your physician on a case-by-case basis to decide the need for PSA. At age 70 and above, PSA testing is discouraged. The American Cancer Society says PSA testing should be discussed if you’re over age 50 and have a life expectancy of 10 years or more. The American Urological Association recommends a PSA every 1-4 years for men ages 45-70. My urologists have ordered PSA tests on me every year since age 60. It has ranged between 4.5-7.1 all this time. I had a negative ultrasound-guided biopsy at age 73. Now, at age 80, I’m told I have a large prostate, but the PSA I have in July will be the last I need.
The knock on PSA testing is the unreliability of the results. The number result doesn’t necessarily correlate with a cancer diagnosis. PSA has a “low positive predictive value.” That means an abnormal PSA is infrequently followed by a positive prostate biopsy. “In men with serum PSA between 3-10 ng/mL the negative biopsy rate is 60-75%…..PSA-based screening has led to an increase in CaP diagnosis and overtreatment because these cancers are “insignificant” (very low chance of mortality from CaP—they are not highly malignant).”
So faced with this conundrum researchers found a genetic biomarker that is far more sensitive and specific than PSA. The marker is called PCA3 and is a segment…. of messenger RNA [found] on a chromosome.” PCA3 is found abundantly “in 95% of all prostate cancers evaluated, and it boasts determining benign cells from cancerous prostate cells with nearly 100% accuracy.” If this test is positive, or elevated, there’s a high percentage chance cancer is present. “It is the most specific CaP biomarker identified to date. Unlike PSA, PCA3 is not affected by age, prostate volume, or other prostate diseases (e.g. prostatitis).”
PCA3 is a genetic test done on urine collected after the patient has a digital rectal exam. During the rectal exam, the prostate is firmly examined with digital pressure applied from top to bottom and from the side to side three times on each lobe. The exam releases prostate cells into the bladder, and they pass out with the urine specimen. The urine specimen is sent to a special lab for analysis. The prostate cells are separated from the urine, and mRNA is, then, extracted from the cells. PCA3 is now determined and is the factor that gives the test a 48%-75% positive predictability. It’s negative predictive value is 74%-90%. The result is reported as a level of mRNA, higher levels being more reliably predictive of cancer. So if the PCA3 is positive, there’s up to a 75% chance a biopsy will show cancer. If the PCA3 is negative, there is a 90% chance there is no cancer, and the biopsy will be negative. When combined with the PSA, diagnostic accuracy increases to 90%. PCA3 is also a good predictor of pre-cancerous lesions, as well. For patients with negative biopsies, an elevated PCA3 alerts the doctor to continue surveillance in this patient.
The PCA3 is manufactured by numerous biotech companies, but I have never heard of it being used routinely in every day practice. I think it is reserved for patients with negative prostate biopsies who have risk factors that place them at a higher level of likelihood of cancer. The normal reference point is a level of 35. Below that number there is little chance of CaP being found, but above 35, CaP needs to be looked for again. It costs between $200 to $450 and is not covered by Medicare. PCA3 is not intended for use in mass screenings for prostate cancer. It’s use is more to reassure the doctor that the patient with a negative biopsy still may have cancer and needs to be re-biopsied at a later time.
Since there is a lot of negative vibe about PSA testing, it’s good to know we have PCA3 testing available to clarify murky situations and give doctors reason to keep a close eye on this individual. The lack of use of this test may be a result of Medicare not covering it, as is the case with any screening-type of test. Knowing about it is helpful.
References: Bourdoumis A, Papatsoris AG, Chrisofos M, Efstathiou E, Skolarikos A, Deleveliotis C. The Novel Prostate Antigen 3 (PCA3) Biomarker. Braz J Urol 2010 Dec;36(6):665-669.
Boehm BE, York, ME, Petrovics G, Kohaar I, Chesnut GT. Biomarkers of Aggressive Prostate Cancer at Diagnosis Int J Mol Sci 2023;24:2185.
Filella X, Foj L, Mila M, Auge JM, Molina R, Jimenez W. PCA3 in the detection and management of early prostate cancer. Tumor Biol 2013 June;34(3):1337-1347.
Day JR, Jost M, Reynolds MA, Groskopf J, Rittenhouse H. PCA3: from basic molecular science to the clinical lab. Cancer Lett 2011 Feb 1;301(1):1-6.


Super article
Thanks